Last updated on Apr 10, 2026

Get the free Batch Manufacturing Record for Tongkat Ali Tablets
We are not affiliated with any brand or entity on this form
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is batch manufacturing record for
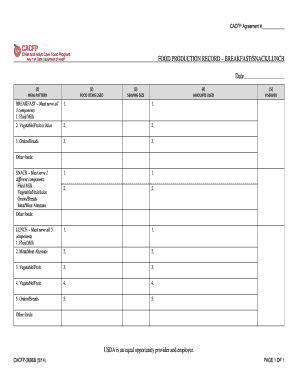
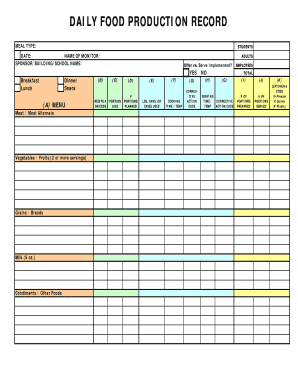
The Batch Manufacturing Record for Tongkat Ali Tablets is a business form used by pharmaceutical manufacturers to document the production process of Tongkat Ali 250mg Tablets and ensure compliance with standard operating procedures.
pdfFiller scores top ratings on review platforms




This program is super user friendly, I am very satisfied with this product, I higgly recommend it!!!!!!
Works great on my PC and I love being able to sign and submit signatures from my tablet. Unfortunately, I'm not able to open my forms on my tablet.
Who needs batch manufacturing record for?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to batch manufacturing record for
What is the Batch Manufacturing Record for Tongkat Ali Tablets?
The Batch Manufacturing Record for Tongkat Ali Tablets is a vital form used in pharmaceutical production to document each step of the manufacturing process. This form captures essential information, including product description, batch size, and storage conditions to ensure accurate tracking of production. It is crucial for maintaining compliance with standard operating procedures (SOPs) within the industry.
The record requires signatures from relevant stakeholders, such as the Production Manager and QA Manager, signifying accountability and adherence to company guidelines. By utilizing this batch manufacturing record template, manufacturers can maintain high standards in product quality and regulatory compliance.
Purpose and Benefits of the Batch Manufacturing Record for Tongkat Ali Tablets
The Batch Manufacturing Record serves critical purposes in the production of Tongkat Ali Tablets. Firstly, it helps track and document the entire manufacturing process, which is essential for maintaining quality control throughout production. This documentation acts as a safeguard against inconsistencies and errors.
-
Enhances compliance with industry regulations.
-
Facilitates accountability by providing a clear record of production activities.
-
Assists in audits and inspections, demonstrating adherence to Good Manufacturing Practices (GMP).
Ultimately, the quality assurance form for tablets underlines the importance of accurate record-keeping in maintaining product integrity and consumer safety.
Key Features of the Batch Manufacturing Record for Tongkat Ali Tablets
This record is designed with several key features, ensuring it meets the diverse needs of pharmaceutical manufacturing. The form contains blank fields for names, signatures, and dates, facilitating easy completion by authorized personnel.
-
Includes checkboxes for specific production steps.
-
Provides clear instructions for users to follow.
-
Can be adapted to meet different production requirements.
Overall, this structure allows for a comprehensive overview while ensuring all necessary components are present for effective record management.
Who Needs the Batch Manufacturing Record for Tongkat Ali Tablets?
The Batch Manufacturing Record is critical for several roles within the pharmaceutical production process. Key personnel who interact with this form include Production Managers and QA Managers, both of whom play vital roles in ensuring compliance and quality assurance.
-
Production Managers are responsible for overseeing the manufacturing process and ensuring that all standards are met.
-
QA Managers must approve the record to confirm that all procedures align with regulatory requirements.
-
Additional personnel, such as Manufacturing Technicians, may also need to reference or sign the record during production.
The involvement of these stakeholders is essential for effective compliance management.
How to Fill Out the Batch Manufacturing Record for Tongkat Ali Tablets Online (Step-by-Step)
Filling out the Batch Manufacturing Record online involves a straightforward process that can be accomplished with an online PDF editor. Follow these steps to ensure accurate completion:
-
Open the digital form in the PDF editor.
-
Enter the product details, including the batch size and storage conditions.
-
Fill in your name and title in the designated fields.
-
Provide the necessary signatures as required.
-
Review the completed form to avoid common pitfalls.
Utilizing visual aids or tips can enhance navigation through the digital form and improve the overall experience.
Digital Signature vs. Wet Signature Requirements
Understanding the differences between digital and wet signatures is crucial for compliance when filling out the Batch Manufacturing Record. A digital signature is often acceptable for most regulations, offering a secure and efficient signing method.
-
Digital signatures can be used for forms that are submitted electronically.
-
Wet signatures may be required for specific regulatory needs or traditional submission methods.
Employing secure signing options within the pdfFiller platform ensures that sensitive data is protected while complying with industry standards.
Submission Methods and Delivery for the Batch Manufacturing Record for Tongkat Ali Tablets
Submitting the completed Batch Manufacturing Record can be done through various methods, making the process flexible. Common submission options include:
-
Online submission via the dedicated portal.
-
Mailing a hard copy to the appropriate regulatory authority.
Each method may have associated fees and processing times. It is essential to confirm your submission and track the status to ensure proper compliance with documentation requirements.
Security and Compliance for the Batch Manufacturing Record for Tongkat Ali Tablets
Security and compliance are paramount when managing sensitive records in pharmaceutical production. pdfFiller employs robust security features, including encryption and compliance with regulations such as HIPAA and GDPR.
-
Ensure proper safeguarding of sensitive information during document handling.
-
Adhere to record retention requirements to maintain compliance.
Implementing best practices for document storage and management will enhance the security of your records.
Sample or Example of a Completed Batch Manufacturing Record for Tongkat Ali Tablets
Providing a sample filled-out Batch Manufacturing Record can serve as a practical reference for users. This example highlights key sections and common entries to guide proper completion.
-
Focus on understanding the rationale behind each component of the form.
-
Use the example as a template rather than something to replicate exactly.
By utilizing samples, users can improve their comprehension of the form's structure and expectations.
Experience Simplified Form Management with pdfFiller
pdfFiller offers an efficient solution for managing the Batch Manufacturing Record. Users can leverage its features for editing, eSigning, and secure sharing of documents.
-
Edit text and images and create fillable forms easily.
-
Benefit from secure document management capabilities.
By utilizing pdfFiller, users can streamline the process of completing forms and enhance overall efficiency.
How to fill out the batch manufacturing record for
-
1.To complete the Batch Manufacturing Record for Tongkat Ali Tablets on pdfFiller, first, access the platform and log into your account.
-
2.Search for the Batch Manufacturing Record template by entering its name in the search bar.
-
3.Once located, open the document to start editing. Review the fields that need completion such as product description and batch size.
-
4.Before filling the form, gather all production-related information such as production steps, storage conditions, and batch details.
-
5.Begin filling in the blank fields systematically, ensuring to enter accurate and detailed information for clarity.
-
6.Utilize pdfFiller's tools to add signatures in the designated lines, ensuring all required personnel such as the Production Manager and QA Manager sign the appropriate sections.
-
7.If necessary, refer to the provided instructions or guidelines within the form for specific details and checkboxes.
-
8.After completing all fields and obtaining the necessary signatures, review the entire document to ensure all information is correct and compliant with SOPs.
-
9.Upon final review, save your work on pdfFiller. You can also download the filled form in PDF format for your records.
-
10.If you need to submit the document, follow the guidelines provided by your organization or regulatory body regarding submission methods or electronic filing.

Who is eligible to complete the Batch Manufacturing Record?
Eligibility to complete the Batch Manufacturing Record typically includes positions such as Production Managers and Quality Assurance Managers who supervise or are involved in the tablet manufacturing process.
Are there any deadlines for submitting the completed form?
Deadlines for submitting the Batch Manufacturing Record may vary based on your company’s protocols or regulatory requirements. Ensure to check internal policies for specific timelines.
How should I submit the completed Batch Manufacturing Record?
Submission procedures for the Batch Manufacturing Record generally involve sharing the completed and signed document with the Quality Assurance department or relevant regulatory bodies, either electronically or in printed form.
What supporting documents are needed with this form?
Common supporting documents may include production logs, raw material specifications, and any other relevant Quality Assurance documents that corroborate the production process outlined in the Batch Manufacturing Record.
What are common mistakes to avoid when filling out the form?
Common mistakes include missing signatures, incomplete fields, and incorrect batch details. Always double-check for accuracy and ensure that all required sections are filled out before submission.
How long does it take to process the Batch Manufacturing Record?
Processing times for the Batch Manufacturing Record can vary based on company regulations and workload within the Quality Assurance team. Typically, review may take anywhere from a few days to over a week.
Is notarization required for the Batch Manufacturing Record?
No, notarization is not required for the Batch Manufacturing Record for Tongkat Ali Tablets, as it is an internal document primarily used for compliance and tracking purposes within pharmaceutical manufacturing.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.